News | June 26, 2023
Edgewise Therapeutics Announces Positive 12-Month Topline Results From The ARCH Open Label Study Of EDG-5506 In Adults With Becker Muscular Dystrophy (BMD)
– Sustained positive trend in North Star Ambulatory Assessment (NSAA) scores relative to BMD natural history –
– Significant decreases in levels of serum creatine kinase (CK) and fast skeletal muscle troponin I (TNNI2), enzyme biomarkers associated with skeletal muscle damage–
– EDG-5506 was well-tolerated at all doses –
– Company continuing to enroll patients with BMD in the CANYON Phase 2 trial which now includes a pivotal cohort called GRAND CANYON –
– Edgewise leadership to discuss ARCH findings on Tuesday, June 27 at 8:30 a.m. Eastern Time at virtual investor event –
BOULDER, Colo.–(BUSINESS WIRE)– Edgewise Therapeutics, Inc., (Nasdaq: EWTX), a clinical-stage biopharmaceutical company focused on developing orally bioavailable, targeted, small molecule therapies for the treatment of devastating muscle disorders, announced today positive 12-month topline results from the ongoing ARCH study, an open label, single-center study assessing the safety, tolerability, impact on muscle damage biomarkers, and pharmacokinetics (PK) of EDG-5506 in adults with BMD. EDG-5506 is an orally administered small molecule designed to prevent contraction-induced muscle damage in dystrophinopathies including BMD and Duchenne muscular dystrophy (DMD).
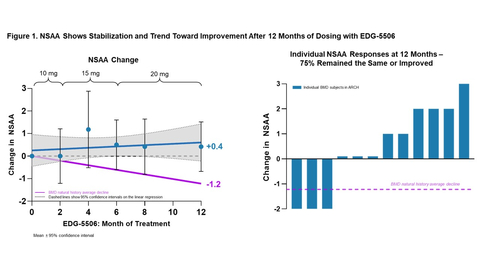
Edgewise Therapeutics Figure 1. NSAA shows stabilization and trend toward improvement after 12 months of dosing with EDG-5506 (Graphic: Business Wire)
Edgewise Therapeutics Figure 1. NSAA shows stabilization and trend toward improvement after 12 months of dosing with EDG-5506 (Graphic: Business Wire)
 The ARCH study is evaluating varying doses of EDG-5506 administered daily over 24 months in 12 adults with BMD. The Company is reporting data at the end of 12 months of treatment with EDG-5506. EDG-5506 was well-tolerated in all participants with no discontinuations or dose reductions due to adverse events.
The ARCH study is evaluating varying doses of EDG-5506 administered daily over 24 months in 12 adults with BMD. The Company is reporting data at the end of 12 months of treatment with EDG-5506. EDG-5506 was well-tolerated in all participants with no discontinuations or dose reductions due to adverse events.
Consistent with prior observations, treatment with EDG-5506 led to significant decreases in key biomarkers of muscle damage. Importantly, CK and TNNI2 were reduced by an average of 37% (p=0.001) and 79% (p<0.0001) from baseline, respectively, at the participants 12-month visit.
After 12 months of EDG-5506 dosing, NSAA scores continued to trend in a positive direction. Nine of the twelve participants showed either a functional improvement (n=6) or exhibited stable disease (n=3) on NSAA relative to their baselines. As seen in Figure 1, NSAA scores show a consistent positive trend that diverges from trajectories observed in the natural history studies reported by Bello et al. (2016)1 and van de Velde et al. (2021)2 in which the yearly decline was -1.2 NSAA points. Overall, one-year functional results were observed to have a +0.4-point improvement on the NSAA compared to the -1.2-point anticipated natural history decline in a population of BMD patients predicted to have a relentless course of disease progression.
The positive results from the 12-month ARCH study support the hypothesis that a reduction in contraction-induced muscle damage in muscular dystrophies, associated with EDG-5506 administration, has the potential to preserve and improve muscle function while preventing disease progression in dystrophinopathies. Observations from ARCH identified key factors, including the optimal dosing strategy of EDG-5506, for the design of a potentially registrational trial. A pivotal cohort, GRAND CANYON, has been added to the CANYON study and is anticipated to begin enrollment in the third quarter of 2023.
“We are quite pleased by the robust and consistent results observed at 12 months with EDG 5506 treatment, which, together with the favorable safety/tolerability profile, are highly encouraging. Moreover, these data have allowed us to quickly pivot our CANYON study to include a potentially registration-enabling cohort,” said Joanne Donovan, M.D., Ph.D., Chief Medical Officer of Edgewise. “We thank the patients, their families, caregivers, and the broader Becker community, who are currently underserved with no available treatment options, for engaging with us on this promising therapy.”
“Becker is a devastating, neuromuscular disease which currently has no approved therapies. In my experience, once symptoms of muscle weakness occur, muscle has already been lost. Once declining, individuals with Becker have a relentless course of disease progression,” said Craig McDonald, M.D., Professor and Chair, Department of Physical Medicine & Rehabilitation, University of California Davis. “I have been closely following the progress of EDG-5506 in the clinic and am excited by the 12-month results and EDG-5506’s potential as a novel therapy for individuals with Becker.”
ARCH Data Review Virtual Investor Event
Members of the Edgewise management team will hold a live webcast on Tuesday, June 27, at 8:30 a.m. ET to discuss the ARCH 12-month data, and will be joined by Dr. Craig McDonald, who will share his perspective of EDG-5506 and BMD. An accompanying slide presentation will also be available. To register for the live webcast and replay, please visit the Edgewise events page.
Further, Edgewise plans to discuss these data at the Parent Project Muscular Dystrophy Annual Conference, June 29 – July 1, 2023.
Edgewise Therapeutics Presentations at Parent Project Muscular Dystrophy Conference
Edgewise Therapeutics’ Chief Medical Officer, Joanne Donovan, M.D., Ph.D., will present at the Parent Project Muscular Dystrophy Annual Conference on July 29, during the “Emerging Therapies in Becker” session at 5:00 pm CT / 6:00 pm ET and on June 30, during the “Research Row: Impacting Downstream” session at 10:45 am CT / 11:45 am ET. Only registered conference attendees can attend the presentations. The Impacting Downstream session will be streamed to the virtual element of the event and both will be available for replay via the conference platform. The Edgewise presentations will be available on the Edgewise website when they are presented.
About the ARCH Open-Label Study
The ARCH open-label study is evaluating EDG-5506 in 12 adult males with BMD. The study is evaluating varying doses of EDG-5506 administered daily over 24 months. Safety, PK, changes in biomarkers of muscle damage such as CK and fast skeletal muscle troponin I, measures of function with NSAA and NSAD, time function tests and patient-reported outcomes are being evaluated. Go to clinicaltrials.gov to learn more about this study (NCT05160415).
About the CANYON Clinical Trial
The CANYON clinical trial is evaluating EDG-5506 in individuals with BMD. CANYON is assessing the effect of EDG-5506 over a 12-month period on safety, PK, biomarkers of muscle damage, such as CK and fast skeletal muscle troponin I, fat fraction as measured by muscle MRI and function in individuals with BMD aged 12 to 50 years. This placebo-controlled trial is anticipated to recruit approximately 32 adults and 18 adolescents at up to 14 sites in the United States, United Kingdom and the Netherlands. The GRAND CANYON cohort has been designed as a potentially registrational cohort with NSAA as the primary endpoint. GRAND CANYON will be a global placebo-controlled study and is anticipated to recruit approximately 120 BMD patients who will receive study drug for 18 months. Go to clinicaltrials.gov to learn more about this trial (NCT05291091).
About Becker Muscular Dystrophy
BMD is a genetic, progressively debilitating, and potentially fatal inherited X-linked neuromuscular disorder. BMD results from mutation of the dystrophin gene yielding unstable and/or dysfunctional dystrophin expression in muscles. Individuals with BMD, typically males, have ongoing muscle fiber (myofiber) degeneration that eventually leads to fibrosis, progressive loss of skeletal muscle function, and that can lead to severe disability and early death. BMD typically presents with juvenile onset of muscle wasting and progressive symmetrical, proximal muscle weakness, calf hypertrophy, activity-induced muscle cramping and elevated CK activity. While the course of BMD is variable, it is unidirectional in terms of the inevitable progressive limb weakness resulting in severe disability. BMD is also associated with early mortality from cardiac disease. The incidence of BMD is approximately 1 in every 18,450 live male births. It is estimated that there are approximately 5,000 individuals with BMD in the U.S., with similar numbers of individuals living with BMD in the EU and UK. Despite the seriousness of the disease, for many with BMD, the disease remains one of considerable unmet medical need as there are no approved therapies.
About EDG-5506
EDG-5506 is an orally administered small molecule designed to prevent contraction-induced muscle damage in dystrophinopathies including DMD and BMD. EDG-5506 presents a novel mechanism of action designed to selectively limit the exaggerated muscle damage caused by the absence or loss of functional dystrophin. By minimizing the progressive muscle damage that leads to functional impairment, EDG-5506 has the potential to benefit a broad range of patients suffering from debilitating rare neuromuscular disorders. It is anticipated to be used as a single agent therapy, but it may also provide an additional benefit in combination with available therapies and therapies currently in development. In August 2021, the U.S. Food and Drug Administration (FDA) granted Fast Track designation to EDG-5506 for the treatment of individuals with BMD.
The Company has completed a Phase 1 clinical trial of EDG-5506 designed to evaluate safety, tolerability, PK and pharmacodynamics of EDG-5506 in adult healthy volunteers (Phase 1a) and in adults with BMD (Phase 1b) (NCT04585464). In ARCH, an open-label, single-center trial (NCT05160415) assessing long-term safety and PK, decreases in biomarkers of muscle damage and trends toward improvement in NSAA have been observed following 12 months of treatment with EDG-5506. CANYON, an ongoing Phase 2 trial (NCT05291091), is assessing safety, PK, biomarkers of muscle damage, fat fraction as measured by muscle MRI and function in participants with BMD aged 12 to 50 years. CANYON now also includes the GRAND CANYON cohort, designed as a potentially registrational cohort with NSAA as the primary endpoint. LYNX, an ongoing Phase 2 trial (NCT05540860), is assessing safety, PK and biomarkers of muscle damage in participants with DMD. The Company is also continuing to recruit the DUNE Phase 2 exercise challenge study, to evaluate the effect of EDG-5506 on biomarkers of muscle damage following exercise in adults with Limb Girdle muscular dystrophy 2i (LGMD2i), BMD or McArdle disease at a single site in Denmark.
About Edgewise Therapeutics
Edgewise Therapeutics is a clinical-stage biopharmaceutical company focused on the discovery, development, and commercialization of innovative treatments for severe, rare neuromuscular and cardiac disorders for which there is significant unmet medical need. Guided by its holistic drug discovery approach to targeting the muscle as an organ, Edgewise has combined its foundational expertise in muscle biology and small molecule engineering to build its proprietary, muscle-focused drug discovery platform. Edgewise’s platform utilizes custom-built high throughput and translatable systems that measure integrated muscle function in whole organ extracts to identify small molecule precision medicines regulating key proteins in muscle tissue. The Company’s lead candidate, EDG-5506, an investigational orally administered small molecule designed to protect injury-susceptible fast skeletal muscle fibers in dystrophinopathies, is advancing in multiple clinical trials in individuals with Duchenne, Becker and Limb Girdle 2I/R9 muscular dystrophies, and McArdle disease. The Company is also advancing EDG-7500, a novel sarcomere modulator for hypertrophic cardiomyopathy, through IND-enabling preclinical development. To learn more, go to: www.edgewisetx.com or follow us on LinkedIn, Twitter and Facebook.
References
[1] Bello L, et al., Functional Changes in Becker Muscular Dystrophy: Implications for Clinical Trials in Dystrophinopathies, Scientific Reports, 2016.
[2] van de Velde et al., Selection Approach to Identify the Optimal Biomarker Using Quantitative Muscle MRI and Functional Assessments in Becker Muscular Dystrophy, Neurology, 2021.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements as that term is defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Statements in this press release that are not purely historical are forward-looking statements. Such forward-looking statements include, among other things, statements regarding the potential of, and expectations regarding Edgewise’s expectations relating to its clinical trials and clinical development of EDG-5506; statements regarding a potentially registrational cohort in the CANYON study and its ability to recruit patients for such study; statements regarding Edgewise’s pipeline of product candidates and programs; and statements by Edgewise’s president and chief medical officer and the Chair, Department of Physical Medicine & Rehabilitation, University of California Davis. Words such as “believes,” “anticipates,” “plans,” “expects,” “intends,” “will,” “goal,” “potential” and similar expressions are intended to identify forward-looking statements. The forward-looking statements contained herein are based upon Edgewise’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those projected in any forward-looking statements due to numerous risks and uncertainties, including but not limited to: risks associated with the process of discovering, developing and commercializing drugs that are safe and effective for use as human therapeutics and operating as an early clinical stage company including the potential for Edgewise’s product candidates to cause serious adverse events; Edgewise’s ability to develop, initiate or complete preclinical studies and clinical trials for, obtain approvals for and commercialize any of its product candidates for muscular dystrophy patients or other patient populations; the timing, progress and results of clinical trials for EDG-5506; Edgewise’s ability to raise any additional funding it will need to continue to pursue its business and product development plans; the timing, scope and likelihood of regulatory filings and approvals; the potential for any clinical trial results to differ from preclinical, interim, preliminary, topline or expected results; Edgewise’s ability to develop a proprietary drug discovery platform to build a pipeline of product candidates; Edgewise’s manufacturing, commercialization and marketing capabilities and strategy; the size of the market opportunity for Edgewise’s product candidates; the loss of key scientific or management personnel; competition in the industry in which Edgewise operates; Edgewise’s reliance on third parties; Edgewise’s ability to obtain and maintain intellectual property protection for its product candidates; general economic and market conditions; and other risks. Information regarding the foregoing and additional risks may be found in the section entitled “Risk Factors” in documents that Edgewise files from time to time with the U.S. Securities and Exchange Commission. These forward-looking statements are made as of the date of this press release, and Edgewise assumes no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements, except as required by law.
Investors & Media
Michael Carruthers
Chief Financial Officer
ir@edgewisetx.com
Source: Edgewise Therapeutics
 The ARCH study is evaluating varying doses of EDG-5506 administered daily over 24 months in 12 adults with BMD. The Company is reporting data at the end of 12 months of treatment with EDG-5506. EDG-5506 was well-tolerated in all participants with no discontinuations or dose reductions due to adverse events.
The ARCH study is evaluating varying doses of EDG-5506 administered daily over 24 months in 12 adults with BMD. The Company is reporting data at the end of 12 months of treatment with EDG-5506. EDG-5506 was well-tolerated in all participants with no discontinuations or dose reductions due to adverse events.